Prof. Dr. Hadi Nur
Modern concepts in catalysis that emerge currently use of the existing basic principles of physics and chemistry of catalytic processes. One of the important catalytic systems is catalysis in the immiscible liquid-liquid system. It has attracted broad interest due to its potential applications as the catalyst system in various chemical reactions for the synthesis of useful chemical products. It realized that the utilization of immiscible liquid-liquid systems is technologically and theoretically relevant. Several catalyst systems, i.e., phase-transfer catalysis (PTC) [1], triphase catalysis (TPC) [2], and phase-boundary catalysis (PBC) [2-8] have been reported in the scientific literature.
The main difference between PTC, TPC, and PBC lies in the type of catalysis. The PTC is a homogeneous catalytic system, while TPC and PBC are heterogeneous catalytic systems that use solid catalysts. Another significant difference is the condition of the reactions. Mechanical agitation, i.e., stirring, is necessary for the PTC and TCP, while for PBC, the reaction is carried out under static conditions without any mechanical agitation. This is an excellent feature of PBC. Based on conceptual design, the PTC and PBC systems are the same. The only difference between TPC and PBC is the type of catalyst used. TPC used organic polymer, and PBC used inorganic porous material. PBC catalyst is designed so that the location of the catalytic active site is located precisely on liquid-liquid interphase. To further clarify this, Fig. 1 shows the differences between PTC, TPC, and PBC.
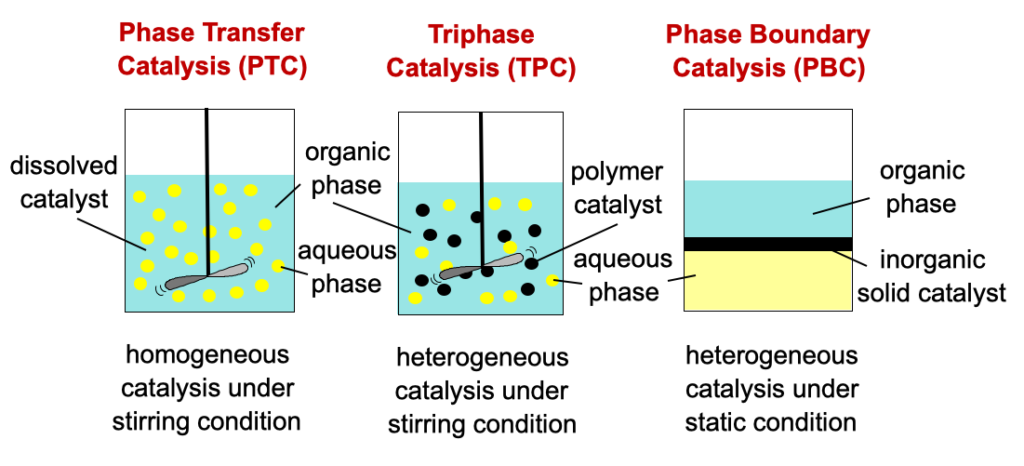
It is rather challenging to compare the catalytic activities and selectivities of PTC, TPC, and PBC because the active sites, reaction types, reactors, and parameters used are different. Unfortunately, there is no research conducted to compare the catalytic performance of PTC, TPC, and PBC. However, it can still be concluded that PTC is a better system in terms of catalytic activity compared to TPC due to the diffusion constraints of TPC.
All the heterogeneous catalysis research has similar issues to be solved that are how to improve the efficiency of adsorption, chemical reaction, and desorption in catalytic cycle processes and how to design an active catalytic site. While the homogeneous catalysis has dominated the development in this research area, it suggests that future research will be in the direction of the synthesis of solid catalysts that possess amphiphilic characters so that they can be positioned at liquid-liquid interphase. Besides, the design of the catalyst needs to pay attention to the location of the active site so that hydrophilic and hydrophobic substrate should easily reach the catalytic active site without limitation of mass transfer. Since many phase-transfer catalyzes (PTC) showed excellent catalytic performance, the catalytic active sites of the phase-transfer catalyst can be mimicked and attached to the amphiphilic solid catalyst. Thus the design of the catalytic active site of solid catalysts and mass transport phenomena of the immiscible liquid-liquid catalysis are the potential topics to be explored in the future.
References
- C.M. Starks, Phase-transfer Catalysis. I. Heterogeneous Reactions Involving Anion Transfer by Quaternary Ammonium and Phosphonium Salts, J. Am. Chem. Soc., 1971, 93, 195-199, DOI: 10.1021/ja00730a033.
- S.L. Regen, Triphase Catalysis, J. Am. Chem. Soc., 1975, 97, 5956-5957, DOI: 10.1021/ja00853a074.
- H. Nur, S. Ikeda, B. Ohtani, Phase-boundary Catalysis: A New Approach in Alkene Epoxidation with Hydrogen Peroxide by Zeolite Loaded with Alkylsilane-Covered Titanium Oxide, Chem. Commun., 2000, 2235-2236, DOI: 10.1039/B006207N.
- H. Nur, S. Ikeda, B. Ohtani, Phase-boundary Catalysis of Alkene Epoxidation with Aqueous Hydrogen Peroxide using Amphiphilic Zeolite Particles Loaded with Titanium Oxide, J. Catal., 2001, 204, 402-408, DOI: 10.1006/jcat.2001.3386.
- S. Ikeda, H. Nur, T. Sawadaishi, K. Ijiro, M. Shimomura, B. Ohtani, Direct Observation of Bimodal Amphiphilic Surface Structures of Zeolite Particles for a Novel Liquid-Liquid Phase Boundary Catalysis, Langmuir, 2001, 17, 7976-7979. DOI: 10.1021/la011088c.
- H. Nur, S. Ikeda, B. Ohtani, Phase-boundary Catalysts for Acid-catalyzed Reactions: The Role of Bimodal Amphiphilic Structure and Location of Active Sites, J. Braz. Chem. Soc., 2004, 15, 719-724. DOI: 10.1590/S0103-50532004000500018.
- S. Ikeda, H. Nur, P. Wu, T. Tatsumi, B. Ohtani, Effect of Titanium Active Site Location on Activity of Phase Boundary Catalyst Particle for Alkene Epoxidation with Aqueous Hydrogen Peroxide, Stud. Surf. Sci. Catal., 2003, 145, 251–254. DOI: 10.1016/S0167-2991(03)80207-0.
- L.S. Yuan, R. Razali, J. Efendi, N.A. Buang, H. Nur, Temperature-controlled Selectivity in Oxidation of 1-Octene by Using Aqueous Hydrogen Peroxide in Phase-boundary catalytic system, App. Catal. A, 2013, 461, 21-25. DOI: 10.1016/j.apcata.2013.04.016.

